Insulin taken by 500,000 could up cancer risk
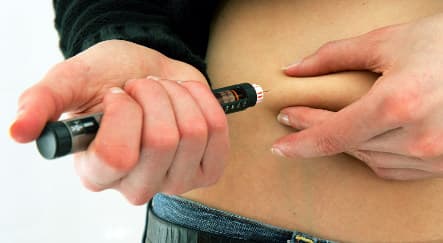
A kind of insulin currently thought to be being taken by around 500,000 German diabetics has been put under suspicion of increasing the risk of cancer by three new studies.
Patients taking the long-acting analogue insulin glargine, sold by Sanofi-Aventis under the name Lantus could have a higher risk of developing cancer than people on other kinds of insulin, according to the studies published in the medical journal Diabetologia.
One of the studies, conducted by the German Institute for Quality and Efficiency in Healthcare (IQWiG), examined the fates of around 130,000 diabetics taking three different kinds of insulin over four years.
A statement from IQWiG said that of those patients taking the more traditional, human insulin, 41 of 1,000 developed cancer in 20 months. But once factors such as age and gender were taken into consideration, within patients taking an average of 10 Glargin units a day, four more per 1,000 developed cancer in that period. Within those on a higher dose of 50 units a day, 13 more cancer diagnoses per 1,000 people were observed.
Peter Sawicki, head of IQWiG and co-author of the study, said, “Our analysis is not clear evidence that glargine encourages cancer. But our study does provoke serious suspicions which should have consequences for the treatment of patients.”
The higher incidence of cancer could be simply a coincidence, but the IQWiG study is being published in Diabetologia with three other studies on the subject, two of which also links glargine with increased cancer rates.
Sawicki said the study was no reason for diabetics taking glargine to suddenly change their drug regime, particularly when their dose is low. But he did say, “If a patient can in any case be just as well treated with human insulin as with glargine, he and his doctor should consider changing. Patients with an increased risk of cancer should use human insulin rather than glargine when somehow possible.”
Glargine has been available in Germany since 2000, promoted with the promise that it does not have to be injected at exactly the same time each day, nor be adjusted according to what the patient has eaten. It is an insulin analogue – an artificial molecule – rather than a human insulin created by the body.
There has been some discussion among scientists that such analogue insulins could, under certain circumstances, promote the growth of cancer cells more strongly than human insulin.
“These clues are discussed by experts, but they have never been dispelled by suitable studies,” said Sawicki.
Der Spiegel magazine reported that up to 500,000 Germans are currently taking glargine and that analogue insulin generally costs between 30 and 60 percent more than human insulin.
The magazine quoted Jean-Pierre Lehner, responsible for medicine security at Sanofi-Aventis, saying the firm did not feel that these latest studies were significant.
“Based on extensive clinical studies with more than 70,000 patients and 24 million patient-years of treatment experience, Sanofi-Aventis continues to regard Lantus as safe,” he told the magazine.
Der Spiegel also reported that Lantus is the company’s second strongest product, expected to bring in more than €3 billion globally this year.
Comments
See Also
Patients taking the long-acting analogue insulin glargine, sold by Sanofi-Aventis under the name Lantus could have a higher risk of developing cancer than people on other kinds of insulin, according to the studies published in the medical journal Diabetologia.
One of the studies, conducted by the German Institute for Quality and Efficiency in Healthcare (IQWiG), examined the fates of around 130,000 diabetics taking three different kinds of insulin over four years.
A statement from IQWiG said that of those patients taking the more traditional, human insulin, 41 of 1,000 developed cancer in 20 months. But once factors such as age and gender were taken into consideration, within patients taking an average of 10 Glargin units a day, four more per 1,000 developed cancer in that period. Within those on a higher dose of 50 units a day, 13 more cancer diagnoses per 1,000 people were observed.
Peter Sawicki, head of IQWiG and co-author of the study, said, “Our analysis is not clear evidence that glargine encourages cancer. But our study does provoke serious suspicions which should have consequences for the treatment of patients.”
The higher incidence of cancer could be simply a coincidence, but the IQWiG study is being published in Diabetologia with three other studies on the subject, two of which also links glargine with increased cancer rates.
Sawicki said the study was no reason for diabetics taking glargine to suddenly change their drug regime, particularly when their dose is low. But he did say, “If a patient can in any case be just as well treated with human insulin as with glargine, he and his doctor should consider changing. Patients with an increased risk of cancer should use human insulin rather than glargine when somehow possible.”
Glargine has been available in Germany since 2000, promoted with the promise that it does not have to be injected at exactly the same time each day, nor be adjusted according to what the patient has eaten. It is an insulin analogue – an artificial molecule – rather than a human insulin created by the body.
There has been some discussion among scientists that such analogue insulins could, under certain circumstances, promote the growth of cancer cells more strongly than human insulin.
“These clues are discussed by experts, but they have never been dispelled by suitable studies,” said Sawicki.
Der Spiegel magazine reported that up to 500,000 Germans are currently taking glargine and that analogue insulin generally costs between 30 and 60 percent more than human insulin.
The magazine quoted Jean-Pierre Lehner, responsible for medicine security at Sanofi-Aventis, saying the firm did not feel that these latest studies were significant.
“Based on extensive clinical studies with more than 70,000 patients and 24 million patient-years of treatment experience, Sanofi-Aventis continues to regard Lantus as safe,” he told the magazine.
Der Spiegel also reported that Lantus is the company’s second strongest product, expected to bring in more than €3 billion globally this year.
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.